- (260) 918-3161
- Bronson, MI 49028
- LPA Admin Login
- Audit Insight
Integrated Real-Time Solutions
High Impact, Low Cost
Understand Your High Risk Areas
Customizable Features
Specific to Your Needs!
Loading color scheme
Insight Lean Solutions is a 100% female owned business, we offer manufacturing audit management software designed to eliminate administrative waste while effortlessly exceeding any customer Layered Process Audit requirements. LPA Admin is easy to use and provides real-time access to critical production process information. Take proactive control of your manufacturing processes.

LPA Admin is designed to manage all your manufacturing audit needs. Our customers can utilize LPA Admin for up to 8 different audits types (i.e., Process, Safety, Environmental, etc.) all in one mobile- responsive application. You can seamlessly transition your organization to paperless auditing through the use of company issued mobile devices. You also get the added benefit of real-time data, automated notifications, and cost savings on paper supplies and Administration time/labor.

Each user of LPA Admin has their own report page and the ability to completely customize their reports to the data they want to see. LPA Admin utilizes Power Bi embedded in our application. This allows our customers to create a quality focused, data-driven culture across your organization. Or you can create a standard reporting format that can be distributed to your team for analysis and report out. This allows everyone in your Leadership to have immediate insights and take quick action.
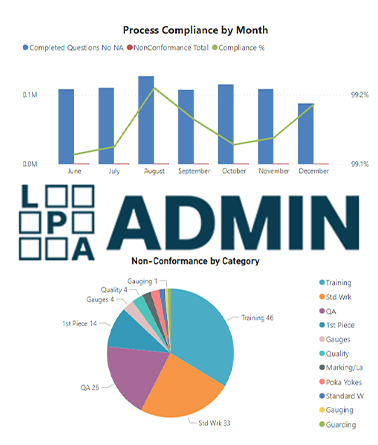
We believe our reputation and growth will move in the same direction as the quality of service our customers are receiving. Our goal is to give them impeccable service. This is why we provide 24/7 customer service to all LPA Admin Administrators at no additional cost. With a team of manufacturing professionals available at all times, we walk alongside our customers from beginning to end; giving you more than just Technical Support, we are available to discuss quality principles, best practices, CQI-8 requirements, Customer Specifics, and assist you with meeting all requirements for your registering body.


Manufacturing Audit Management
Enhance Your Competative Advantage

Don't Make These Mistakes

Lower your annual fee per site subscription!

How Can We Help You?

Ask about a 30 day trial & a live demo.

Layered Process Audits (LPA), by design, are simple, short, and repetitive verifications of process control and conformance. The layered structure makes LPAs an instrumental tool for...

As registrars and customers increasingly place focus on identifying process risks, controlling "at risk" processes, and monitoring those controls, many companies find themselves looking for

CEO
As the Owner & CEO of Insight Lean Solutions, Jamie Noe leads our team with 20 years of experience in manufacturing quality in various commodities
She is a strong believer in Continuous Learning. Jamie has trained her staff and team members on Quality Principles such as Effective Auditing, Problem Solving, Continuous Improvement, and AIAG Core Tools
She continues to strive towards improved quality and efficiency in our applications, as well as impeccable customer support. She is continually monitoring manufacturing trends and assisting in developing training and products that support our client’s needs.